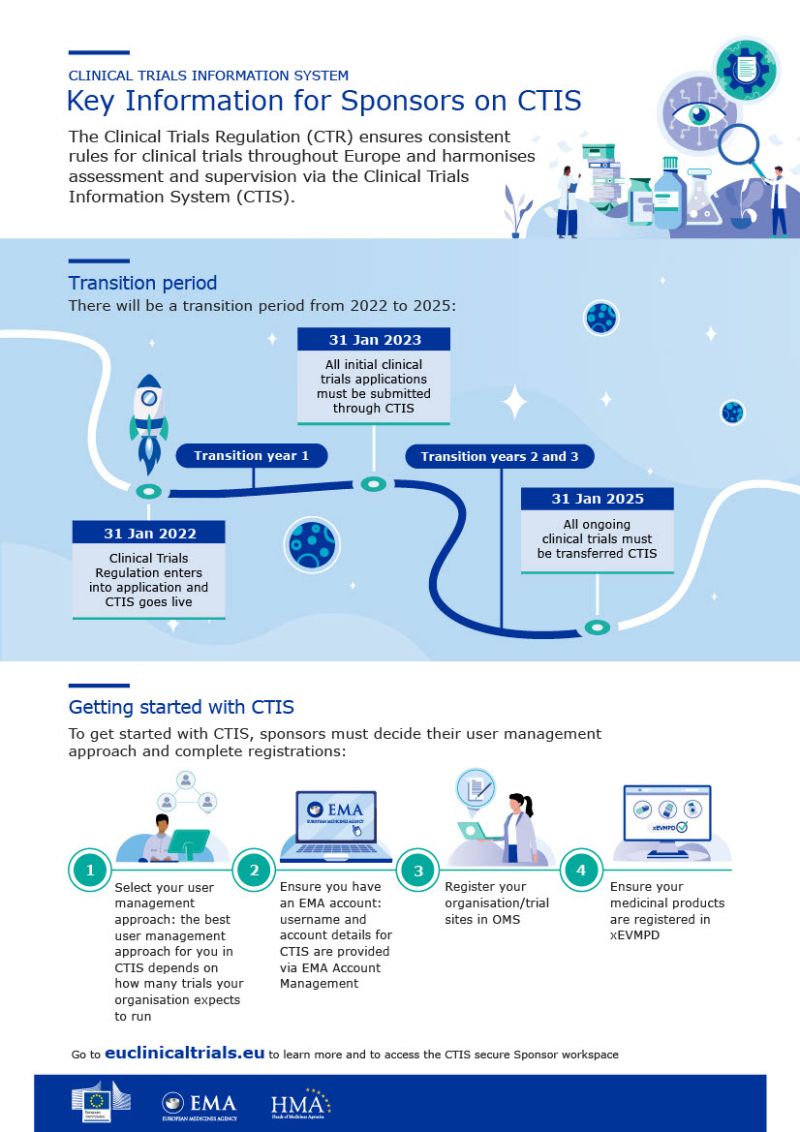
Mandatory Use of Clinical Trial Information System (CTIS) for Initial Clinical Trial Applications in the EU - Avoca, a WCG company

Managing the New EU Clinical Trials Regulation 536/2014 – Guidance for Navigating the Clinical Trial Information System (CTIS)